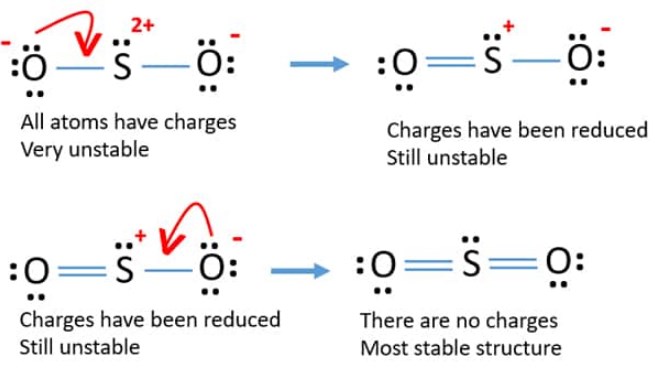
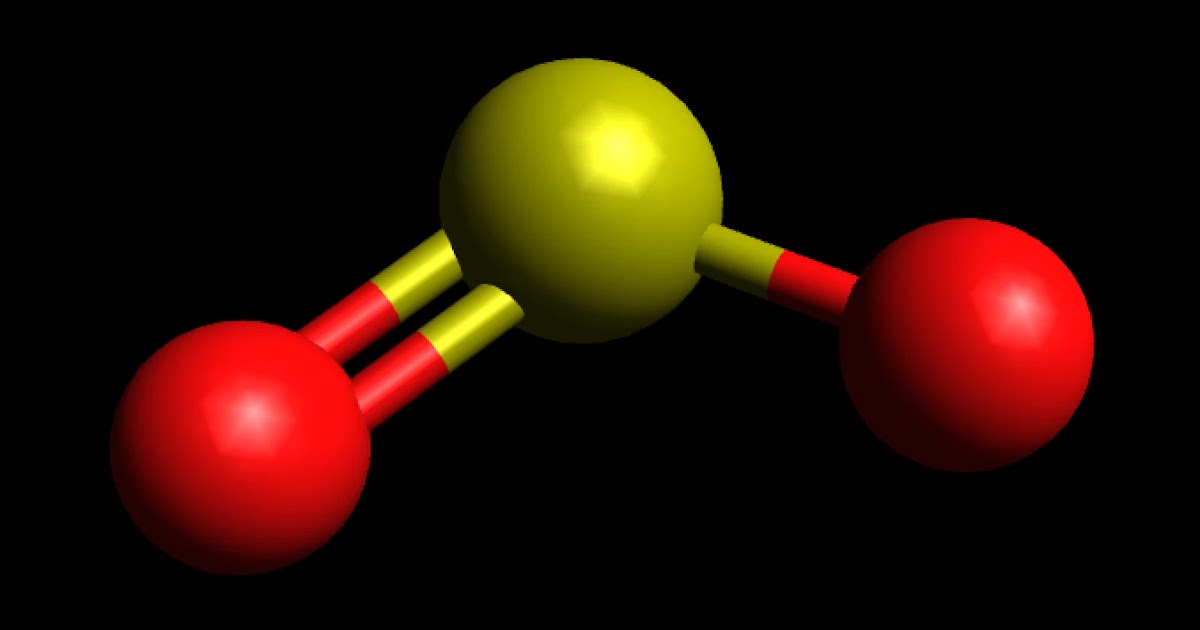
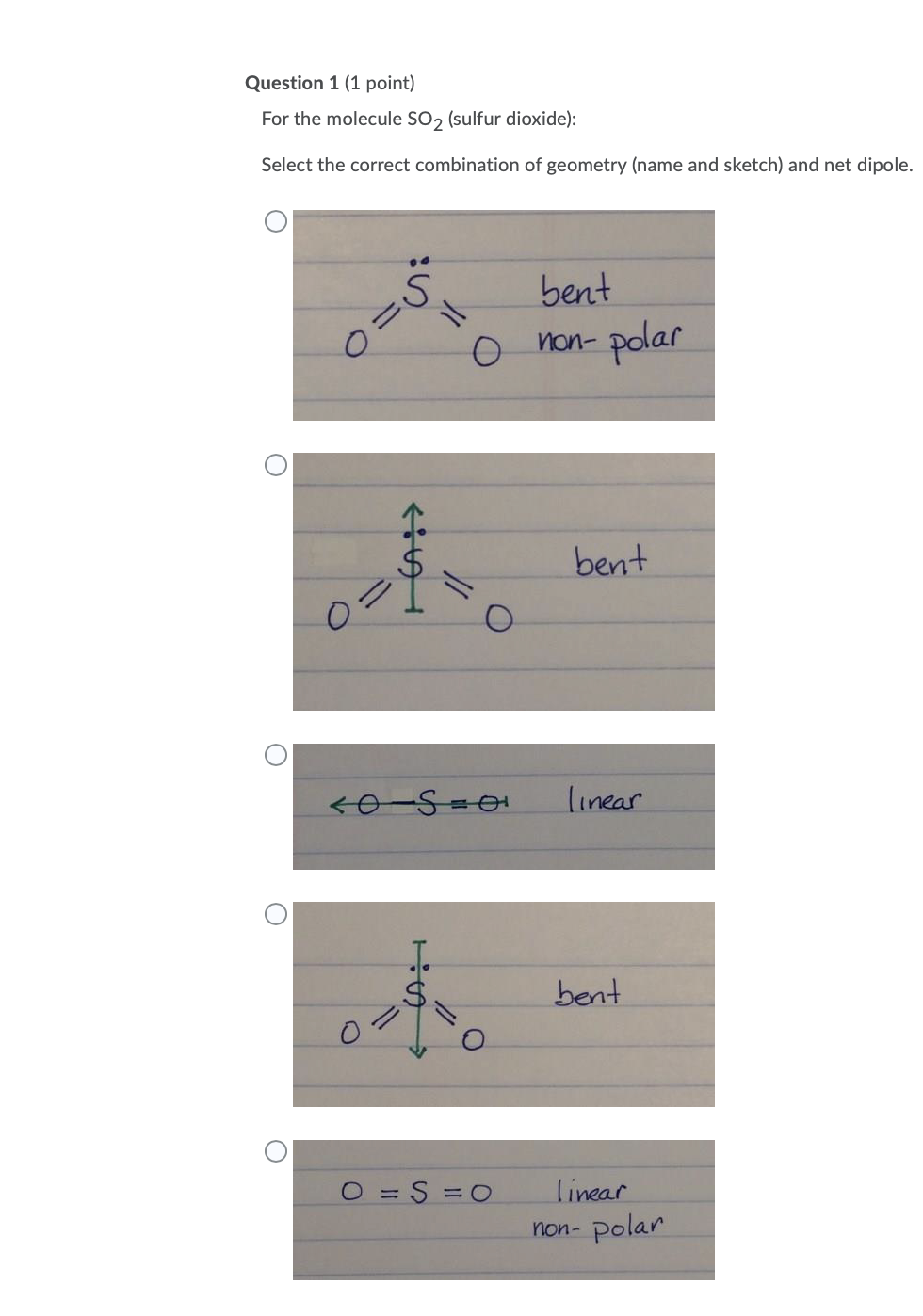
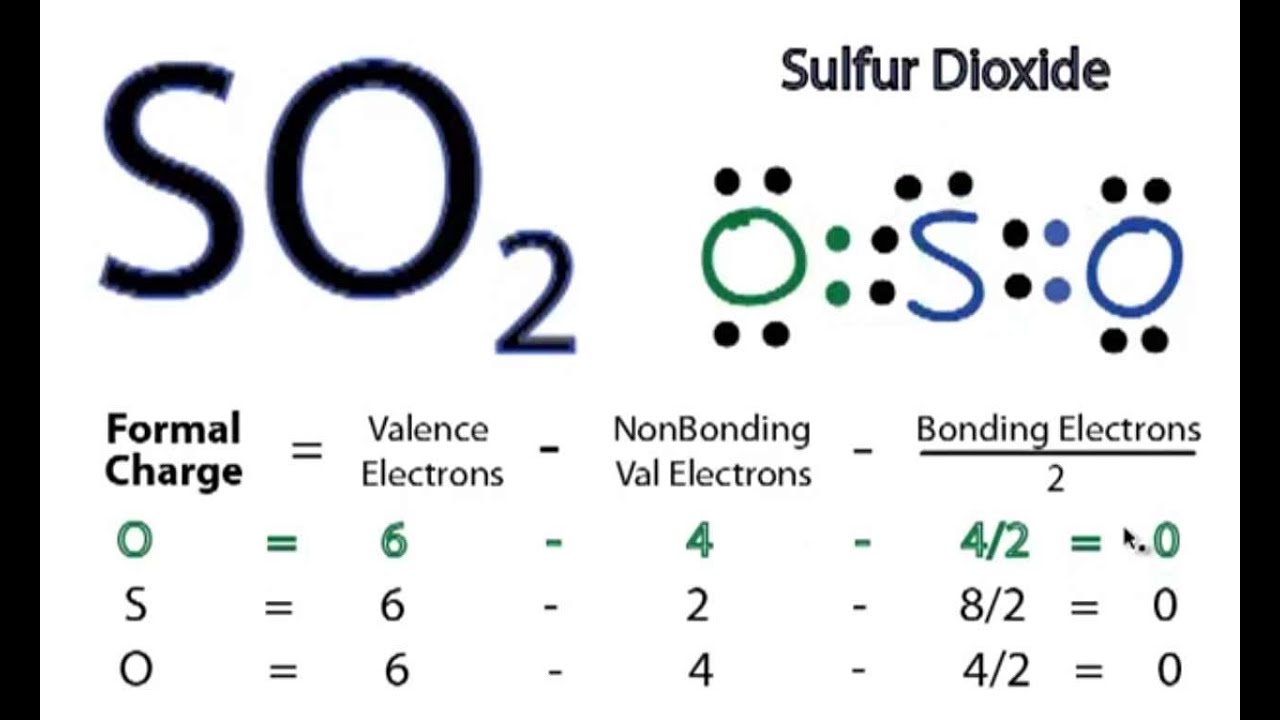
CO2 and SO2 both are triatomic molecules; but there is a big difference in their dipole moment why - Chemistry - Chemical Bonding and Molecular Structure - 6914070 | Meritnation.com
A) Draw lewis structures for CO2, SO2, AND NO3-. B) Give the electron pair geometry and the molecular geometry of the three species from part A) according to VSEPR. C) Are CO2